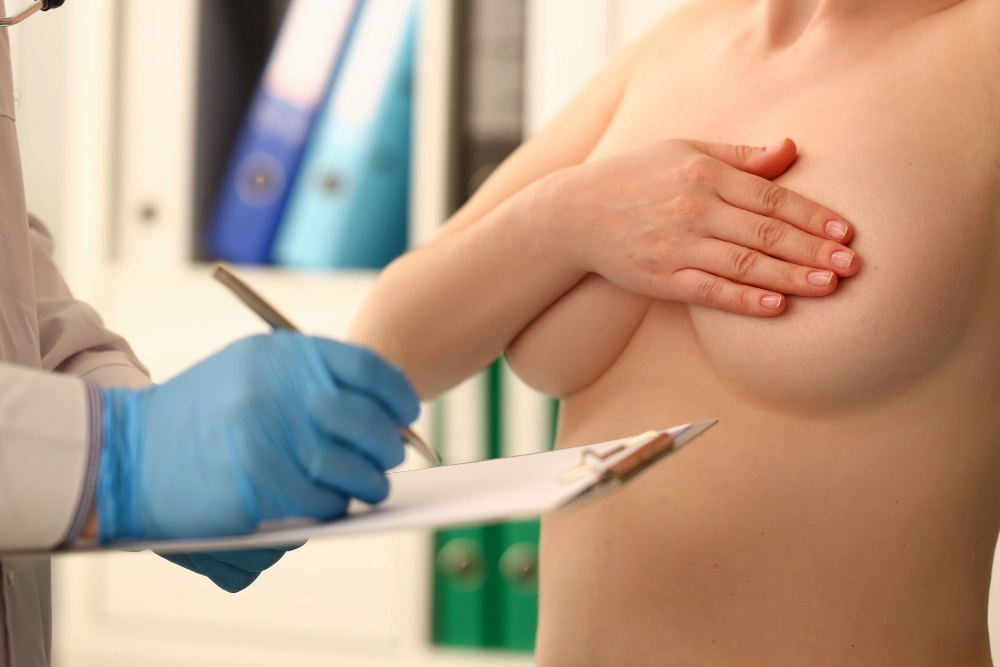
Hyaluronic acid is an authorized material that surgeons use as a filler in various soft parts of the body, such as the buttocks, the breast or specific areas of the face. Now then, is this hyaluronic acid breast treatment recommended? We clarify it for you.
The Spanish Agency for Medicines and Health Products (Aemps) issued a statement in which it reported the «restriction of the use of the injectable filler implant Macrolane [one of the brands under which hyaluronic acid is marketed] for increasing breast volume for aesthetic purposes, because it may interfere with early breast cancer detection techniques». Without the matter posing a serious problem, since the material is perfectly approved and has been applied frequently in all kinds of interventions, surgeons insist that it is still used in many clinics as a filler and in that sense it is more prudent to opt for another material. Specialists say that «in some cases it has been proven that hyaluronic acid can mislead the ultrasound specialist when performing a routine mammogram». Therefore, it is possible that this material may hide the presence of a tumor in the breast in certain tests. The product does not present drawbacks when applied to other parts of the body, but it is preferable to avoid it to fill the breast due to the oncological implications described. The French Health Products Safety Agency was the one that reported the problem to the AEMPS, as it did with the rest of the European authorities, and the need to restrict the use of hyaluronic acid breast implants. The decision was motivated by the results of a study that highlighted the inherent risks of injecting hyaluronic acid breasts, as it can interfere «in physical examination tests and in the reading of mammograms for the early detection of breast cancer». In France, the only product affected by the decision is the injectable hyaluronic acid gel Macrolane manufactured by the Swedish company Q-Med AB. However, the Spanish agency warns that in our country there are other similar products that would be subject to the same warning. Following the publication of the study in France, the Swedish health authorities informed the European authorities that Q-Med AB will cancel the marketing of Macrolane for the indication of increasing breast volume. According to the Aemps statement, the company itself, which is a division of Galderma, would inform professionals about its decision not to continue with that indication. The Spanish agency, in addition to recommending in its note to professionals that they do not use the product for that purpose, suggests that women who have been treated with these injectable implants be informed so that they are aware of the risk. If you want more information, contact our team at contacto@drfernandezblanco.com or through our Facebook The plastic surgeon Dr. Alfredo Fernandez Blanco stands out in his branch of medicine, as the best breast surgeon, he is also a pioneer in the so-called secondary surgeries or sequelae. With more than 30 years of experience and thousands of successful cases, he continues to achieve the most natural results that can be expected in a cosmetic surgery operation.What does the Spanish Agency for Medicines and Health Products say about hyaluronic acid in the breasts?
And what about the specialists?
Who discovered these problems of hyaluronic acid breasts?
French study

Dr. Alfredo Fernández Blanco
Breast augmentationCosmetic surgeryFacial and Body Aesthetic MedicineTratamiento Ácido Hialurónico
Learn why the use of hyaluronic acid breasts is not recommended
Discover our updates
Join our newsletter to stay informed about our new treatments, surgeries, and updates about the team.
| Gracias por registrarte en nuestra Newsletter |

Madrid
Nº NICA: CS-05876
Av. de Filipinas 48
Portal Izq. 1er Piso
28003, Madrid
Marbella
Nº NICA: 59902
Av. Severo Ochoa 53
Local 1 y 3
29603, Marbella
Resources



© 2026 Clínica Fernández Blanco.